Ace Bayou Corp., of New Orleans, La., announced the voluntary recall of about 2.2 million bean bag chairs following the deaths of two children. A 13-year old boy from McKinney, Texas died and a 3-year-old girl from Lexington, Ky. died after suffocating from lack of air and inhaling the chair’s foam beads. Both children were found inside the chairs. The zippers on the bean bag chairs can be opened allowing children to crawl inside, become trapped and suffocate or choke on the bean bag chair’s foam beads.
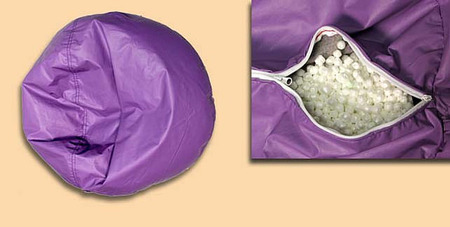
The recalled bean bag chairs have two zippers that can be unzipped and opened, including one of the exterior cover and other directly underneath that zipper. The recalled chairs were sold in a variety of sizes, shapes, colors and fabrics. They include round or L-shaped, vinyl or fabric, and are filled with polystyrene foam beads. The round bean bag chairs were sold in three sizes, 30, 32 and 40 inches in diameter. The L-shaped bean bag chair measures 18 inches wide by 30 inches deep by 30 inches high. “ACE BAYOU CORP” is printed on a tag sewn into the bean bag chair’s cover seam.
The recalled bean bag chairs were sold at Bergner’s, Big Lots, Bon-Ton, Boston Store, Carson’s, Elder-Beerman, Herberger’s, Meijer, Pamida, School Specialty, Wayfair, Walmart and Younkers stores and online at Amazon.com, Meijer.com, Walmart.com and other websites before July 2013 for between $30 and $100.
Consumers should check their bean bag chairs for any zippers that can open, take those that can open away from children immediately and contact Ace Bayou for a free repair kit to permanently disable the zippers so that they cannot be opened.

